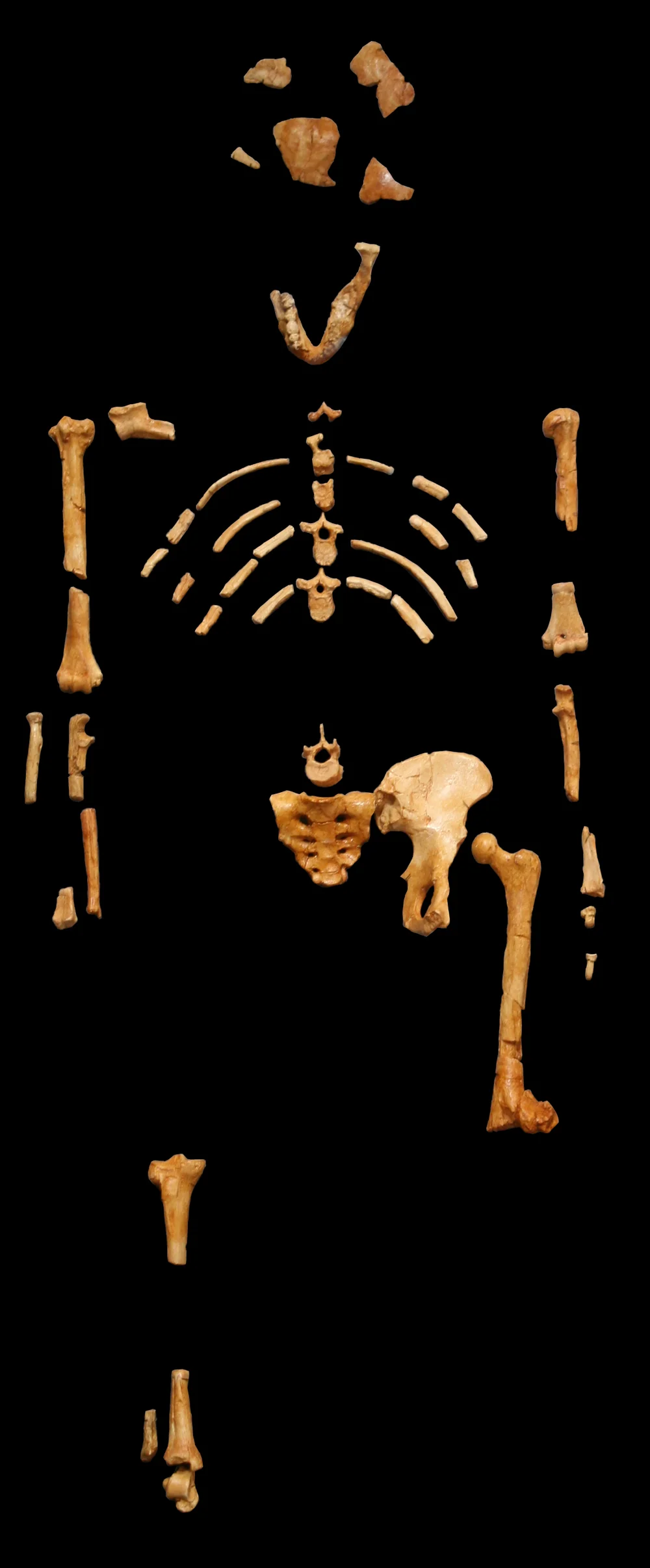
AL 288-1 · Australopithecus afarensis · 3.2 Ma
Lucy: Before the Genus Homo
The Fossil That Rewrote the Story of Walking Upright
The Afar
The Afar Triangle is one of the most geologically violent places on Earth. Wedged between the Ethiopian Plateau, the Somali Plateau, and the Red Sea coast, it sits at the triple junction where three tectonic plates are pulling apart in slow motion, tearing the African continent along a rift that has been widening for roughly 25 million years. The land sinks as the crust thins. In the Danakil Depression, the surface drops to more than 100 meters below sea level, making it one of the lowest and hottest inhabited regions on the planet. Daytime temperatures routinely exceed 50 degrees Celsius. Sulphurous hot springs bubble through salt flats. Basalt flows from Erta Ale, one of the few volcanoes on Earth with a permanent lava lake, creep across a landscape that looks more like another planet than another country.
It is precisely this tectonic violence that makes the Afar indispensable to paleoanthropology. Rifting does two things that fossil hunters require above all else: it creates basins where sediment accumulates and preserves organic remains, and then, millions of years later, it uplifts and erodes those sediments back to the surface. The Hadar Formation, exposed in the badlands along the lower Awash River in Ethiopia’s Afar Region, is one such gift of deep time. Its strata span roughly a million years of Pliocene history, from about 3.8 to 2.9 million years ago, and within those layers lies one of the richest records of early hominin life ever assembled.

The Afar Depression as seen from the ASTER satellite instrument. Three tectonic plates diverge here, creating the rift system that preserved millions of years of sedimentary history.
But 3.2 million years ago, the Hadar region looked nothing like the blasted furnace it is today. Paleoenvironmental reconstructions, built from fossil pollen, stable isotope ratios in ancient soil carbonates, and the species composition of fossilized animal communities, paint a picture of a landscape in mosaic. A paleo-Awash river system, fed by seasonal rains from the Ethiopian highlands, wound through the lowlands carrying silt that built broad floodplains on either side.Gallery forests—ribbons of closed-canopy woodland that hug riverbanks in otherwise open country—lined the channel, providing shade, fruit, and refuge from predators. Beyond the gallery forests, the land opened into mixed grassland and scrubland, with patches of Acacia woodland and seasonal wetlands where the river overflowed during the rains.
This was not a single habitat but a patchwork, and its occupants reflected that diversity. Fossilized bones from the Hadar Formation include bovids (ancestral antelopes), suids (ancient pigs), proboscideans (elephant relatives), hippopotamids, crocodilians, various cercopithecid monkeys, and—critically—hominins. The same sediments that preserved a three-toed horse also preserved the partial skeleton of a small-bodied, upright-walking primate that would, half a century later, become the most famous fossil in the world.

The Hadar badlands today. Millions of years of sedimentary history are exposed in the eroded gullies and ridgelines of this arid landscape along the lower Awash River.
What gives the Hadar Formation its extraordinary chronological precision is volcanic ash. The tuff bands—layers of fine-grained volcanic debris ejected during eruptions from the nearby rift volcanoes—are scattered through the sedimentary column like bookmarks. Each tuff contains radioactive potassium-40, which decays into argon-40 at a known rate. By measuring the ratio of these isotopes in a tuff sample, geochronologists can determine when the eruption occurred, and therefore the age of the sediments immediately above and below it. This technique, known as potassium-argon dating (and its more refined descendant, argon-argon dating), is the reason we can say with confidence that the skeleton catalogued as AL 288-1 is approximately 3.18 million years old. She was buried in the Denen Dora Member of the Hadar Formation, bracketed above and below by datable tuff horizons.
The formation is divided into four members, stacked oldest to youngest, each recording a different chapter in the environmental history of the Hadar basin. The Basal Member preserves a deep lake. The Sidi Hakoma Member records the transition to a river floodplain, and it is here that the first Australopithecus afarensis fossils appear. The Denen Dora Member, thinner and finer-grained, represents a period of marshy floodplain—and it is in this member that Lucy was found. Above it, the Kada Hadar Member records a shift to a higher-energy braided river system, with coarser sands and the last of the Hadar’s afarensis fossils.
Stratigraphic Column — Hadar Formation
D11: Hadar Formation Stratigraphic Column
Four members spanning ~3.80–2.95 Ma. Tuff bands provide radiometric age anchors. The Denen Dora Member (highlighted) yielded AL 288-1.
Understanding this stratigraphic context is essential to understanding Lucy. She is not a fossil plucked from undifferentiated rock. She is embedded in a precisely dated, environmentally characterized, faunal-community-documented slice of the Pliocene. The sediment that buried her tells us what the world around her looked like. The animal bones found in the same deposits tell us who her neighbors were. The tuff that brackets her tells us when she lived. Before the first bone of AL 288-1 was ever described anatomically, the geology had already established the stage.
Key Fossil Sites
Australopithecus afarensis and Related Hominin Sites
Major localities across eastern and southern Africa where A. afarensis and contemporary hominin species have been recovered.
| Site | Lat | Lon | Key Specimens | Age |
|---|---|---|---|---|
| Hadar (Lucy) | 11.10 | 40.58 | AL 288-1, AL 333 | 3.4–2.9 Ma |
| Dikika (Selam) | 11.08 | 40.58 | DIK-1-1 | ~3.3 Ma |
| Laetoli | -3.22 | 35.35 | LH 4, footprints | 3.7–3.5 Ma |
| Sterkfontein | -26.02 | 27.73 | Sts 5, StW 573 | 3.7–2.0 Ma |
| Lomekwi | 3.95 | 35.85 | KNM-WT 40000 | 3.5–3.3 Ma |
| Woranso-Mille | 11.37 | 40.45 | BRT-VP-3/1 | 3.5–3.3 Ma |
| Ledi-Geraru | 11.12 | 40.45 | LD 350-1 | ~2.8 Ma |
| Bouri | 10.27 | 40.38 | BOU-VP-12/130 | ~2.5 Ma |
November 24, 1974
By late November, the field season at Hadar was winding down. Donald Johanson, a young paleoanthropologist from Case Western Reserve University, and his graduate student Tom Gray had spent weeks surveying the eroded badlands of locality 162, cataloguing fossil fragments of ancient fauna—an antelope jaw here, a horse tooth there. The work was painstaking and, most days, uneventful. The Afar heat was relentless. By early afternoon, the air above the exposed sediment shimmered, and the impulse to retreat to camp was strong.
On this particular morning, Johanson and Gray drove out to a different area—Hadar locality 288, an unremarkable gully system that had not yielded anything significant in previous surveys. They arrived before the worst of the heat and began the slow, methodical walk-through that constitutes surface survey: eyes down, scanning the ground for anything that looks organic against the mineral background of sand and silt. Fossil bone, mineralized and heavy, catches light differently than rock. After years of field experience, the eye learns to find it. But most of the time, there is nothing to find.
They mapped the area through the morning. By midday, with the temperature climbing past 40 degrees Celsius, Johanson suggested heading back. But something drew him to take one more look at a small gully they had already passed. It was there, lying on the surface among the eroded pebbles and sun-baked silt, that he spotted what looked like a piece of arm bone—a fragment of a proximal ulna. The shape was right. The size was small but not too small. It was hominin.
And then there was more. A few meters away, a fragment of the back of a skull. Then a piece of femur. Then vertebrae. Rib fragments. A piece of pelvis. The bones were not scattered randomly over a wide area, as they would be if a carnivore had dismembered a carcass and dispersed the remains. They were concentrated, clustered in the sediment of a single eroding slope, as though they had been buried together and were only now washing out. Johanson and Gray stood in the gully, looking at the accumulating evidence on the ground around them, and began to understand what they were seeing: not a fragmentary jaw, not a single tooth, not an isolated limb bone, but pieces of a single skeleton.
This almost never happens. The hominin fossil record is overwhelmingly composed of fragments—isolated teeth, broken jawbones, partial crania. The chances of any individual dying in the right place, being buried quickly enough to escape scavengers and weathering, remaining undisturbed for millions of years, and then eroding back to the surface at the exact moment and location where a trained scientist happens to be walking are astronomically small. To find multiple elements of a single individual together—enough to reconstruct a body, to determine sex, to assess stature, to analyze gait—was something that had essentially not happened before for a hominin this old.
“We realized this was part of a skeleton, something only rarely found in human evolution. Normally, we’re lucky to find a jawbone, a few teeth. Here, we had a knee, ribs, vertebrae, part of a pelvis—all from a single individual.”
— Donald Johanson, recounting the discovery of AL 288-1
They marked the site, collected the surface finds, and drove back to camp in a state of barely contained exhilaration. Word spread through the expedition within minutes. That evening, as the team gathered to celebrate what was already clearly the most important find of the season—possibly the most important find of their careers—someone put a Beatles tape on the camp’s portable cassette player. “Lucy in the Sky with Diamonds” drifted through the desert night air. Someone, in the euphoria of the moment, called the skeleton Lucy. The name stuck, the way nicknames do when they are born at precisely the right emotional pitch. The formal catalogue number would be AL 288-1—Afar Locality 288, specimen 1—but to the world, she would always be Lucy.

AL 288-1 — “Lucy.” Approximately 47 bone fragments representing roughly 40% of a single adult female Australopithecus afarensis, recovered from the Denen Dora Member of the Hadar Formation, Ethiopia.
Over the following three weeks, the team returned to locality 288 day after day, sieving the sediment, crawling on hands and knees across the slope, recovering every fragment they could find. The bones were fragile and mineralized, stained the dark brown of the surrounding sediment. Some were barely larger than a fingernail. Each piece was plotted on a site map, bagged, and numbered. By the end of the excavation, they had recovered several hundred fragments that, when assembled, represented the partial skeleton of a single small-bodied, adult hominin. No duplicate elements were found—no second left femur, no extra mandible—confirming that all the fragments belonged to one individual.
The pelvis told them the individual was female. The degree of dental wear and the state of epiphyseal fusion told them she was an adult, probably in her twenties. The overall morphology—small brain, large face, curved finger bones, but a pelvis and femur adapted for upright walking—placed her firmly outside the genus Homo. She was something older, something different, something that forced a rethinking of the human family tree that is still ongoing half a century later.
Forty Percent
The human skeleton contains 206 bones. Of Lucy’s skeleton, 47 elements were recovered—approximately 23% by raw count. But that number is deceptive. Many of the “missing” bones are small, repetitive elements: individual hand phalanges, foot bones, wrist carpals. In terms of the major functional regions of the body—the skull, the vertebral column, the rib cage, the pelvis, the upper and lower limbs—Lucy is approximately 40% complete. At the time of her discovery in 1974, this made her the most complete early hominin skeleton ever found, by a considerable margin.
To appreciate what this means, you have to understand the baseline. Before Lucy, the hominin fossil record older than two million years consisted almost entirely of cranial fragments, isolated teeth, and occasional limb bones. The famous Taung Child, discovered in 1924, was a face and endocast—extraordinary, but it told you about the head, not the body. The robust australopithecines from Olduvai Gorge and Swartkrans were known from skulls and jaws. Postcranial bones—the skeleton below the skull—were rare, fragmentary, and usually impossible to assign to a specific individual. You might have a femur from one site and a pelvis from another, with no way to know whether they represented the same species, the same sex, or the same body size.
Lucy changed that. Because her bones came from a single individual, every measurement could be cross-referenced with every other measurement. Her femur length could be compared to her humerus length to calculate an intermembral index—the ratio of arm to leg length that distinguishes climbers from walkers. Her pelvic dimensions could be correlated with her femoral anatomy to reconstruct her gait. Her vertebral column, though incomplete, preserved enough lumbar vertebrae to assess whether she had the lordotic curvature that bipeds require for balance. For the first time, paleoanthropologists could study an early hominin not as a collection of isolated parts but as an integrated organism—a body that had moved through the world in a specific way.

The recovered elements of AL 288-1 laid out in anatomical position. The brown-stained bones represent approximately 40% of the functional skeleton of an adult female Australopithecus afarensis.
The distribution of preserved elements is itself informative. The pelvis is nearly complete—a stroke of extraordinary luck, since the pelvis is the single most diagnostic element for determining both locomotion and sex. Both femora survived, along with portions of the tibia and fibula, giving a nearly complete picture of the lower limb. The upper limbs are also well represented: both humeri, a left ulna, and a right radius. Several vertebrae from the cervical, thoracic, and lumbar regions were recovered, along with multiple rib fragments. The cranium is more fragmentary—a few skull vault pieces and a partial mandible with teeth—but enough to estimate cranial capacity and dental morphology.
What is missing is also telling. The hands and feet are poorly represented, preserving only scattered phalanges and a few metacarpals and metatarsals. This is not unusual in taphonomic terms—small bones are more easily carried away by water or scavengers, and more easily overlooked during excavation—but it is frustrating, because hand and foot morphology are central to the ongoing debate about how much time Lucy’s species spent in the trees. The curved phalanges that were recovered have fueled decades of argument. Were they functional adaptations for climbing, retained from an arboreal ancestor and still in active use? Or were they evolutionary baggage—ancestral features that had not yet been erased by natural selection because there was no cost to keeping them?
The skeleton’s preservation also raised questions about how she died and how she came to be buried. The bones show no carnivore tooth marks, no evidence of gnawing or scattering by predators. This suggests she was buried relatively quickly after death, perhaps by a seasonal flood depositing silt over her remains on the Denen Dora floodplain. The question of what killed her—whether she fell from a tree, as one dramatic 2016 hypothesis proposes, or simply died and was buried by ordinary geological processes—remains one of the most debated topics in the Lucy literature.
Skeletal Inventory
D7: Skeletal Completeness of AL 288-1
47 of 206 skeletal elements recovered (~40% functional completeness). Select a body region to see preservation details and ongoing debates.
Despite the gaps, the skeleton that Johanson and his team pieced together remains one of the most important single fossils in the history of the discipline. In the decades since her discovery, other early hominins have been found that rival or exceed her completeness—the 4.4-million-year-old ARA-VP-6/500 (“Ardi”), the 3.3-million-year-old DIK-1-1 (“Selam”), the 3.67-million-year-old StW 573 (“Little Foot”). But in 1974, nothing remotely comparable existed. Lucy was, for years, the only window into what an early hominin body actually looked like as a whole. That window opened onto a creature that walked upright but had a brain the size of a chimpanzee’s, arms proportioned somewhere between ape and human, and a pelvis that told a story the scientific community was not yet ready to fully absorb.
Built to Walk
The argument that Lucy walked upright does not rest on a single bone or a single measurement. It rests on an integrated suite of anatomical features, each of which independently points toward obligate bipedalism—a body built not merely to walk on two legs occasionally, as chimpanzees sometimes do, but to walk on two legs as the primary and habitual mode of locomotion. The case was made most forcefully by C. Owen Lovejoy, a functional morphologist at Kent State University, who analyzed Lucy’s postcrania in a series of landmark papers through the 1970s and 1980s. His argument was anatomical, biomechanical, and comparative, and it changed the way the field understood the origins of upright walking.
The pelvis is the smoking gun. In quadrupedal apes, the ilium—the large, wing-shaped bone that forms the upper part of the pelvis—is tall and narrow, oriented in the sagittal plane (front to back). This configuration is efficient for a body that is held horizontally: the gluteal muscles, attached to the outer surface of the ilium, act primarily as hip extensors, pulling the thigh backward during climbing and quadrupedal walking. In humans, the ilium has been dramatically reshaped. It is short and broad, flared laterally, reorienting the gluteal musculature from extensors to abductors. This is the critical biomechanical shift. When you walk upright, you spend half of each stride balanced on one leg. Without strong hip abductors—muscles that pull laterally to prevent the pelvis from dropping on the unsupported side—you would topple sideways with every step. The short, broad human ilium is the structural solution to this problem.
Lucy’s pelvis is short and broad. Not as short or as broad as a modern human’s—she retains some ancestral features, including a relatively wider pelvic inlet—but unmistakably reorganized for bipedal locomotion. The iliac blade is laterally flared, the sacroiliac joint is broadened, and the attachment sites for the gluteus medius and gluteus minimus—the primary hip abductors—are repositioned in a way that only makes biomechanical sense if the owner of this pelvis habitually walked on two legs. Lovejoy was emphatic on this point: the pelvis of AL 288-1 is a bipedal pelvis. There is no quadrupedal reading of this anatomy that is mechanically viable.
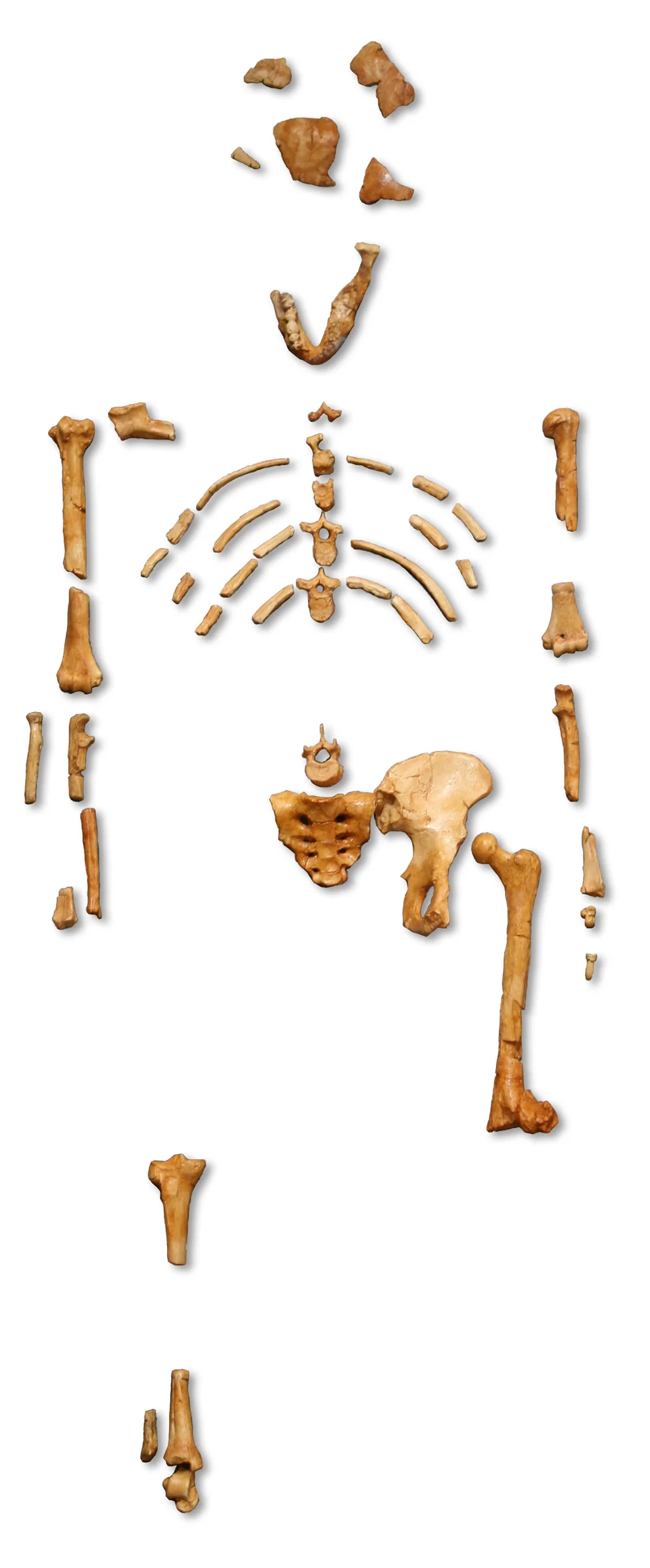
Reconstruction of Australopithecus afarensis based on AL 288-1 and other Hadar specimens. The short, broad pelvis and angled femur are visible indicators of habitual bipedal locomotion.
The femur tells the same story from a different angle—literally. In apes that walk quadrupedally, the femoral shaft drops nearly vertically from the hip to the knee. The bicondylar angle—the angle between the femoral shaft and a line perpendicular to the knee joint surface—is close to zero. In humans, the femur angles inward from hip to knee, creating a bicondylar angle of approximately 9 to 11 degrees. This valgus angle brings the knees closer to the body’s midline, directly beneath the center of gravity, so that each step does not require a lurching lateral weight shift. Lucy’s bicondylar angle is approximately 9 degrees—well within the modern human range, and dramatically different from any quadruped.
Above the pelvis, further evidence accumulates. Lucy’s foramen magnum—the hole in the base of the skull through which the spinal cord passes—is positioned anteriorly, beneath the skull rather than behind it. In quadrupeds, the foramen magnum faces posteriorly, because the skull is held in front of the body and the spine enters from behind. In bipeds, the skull sits atop the spine, and the foramen magnum has migrated to the base. Lucy’s cranial base, though fragmentary, preserves enough to confirm this anterior positioning. Her head sat on top of her spine, not in front of it.
The vertebral column adds more. Several of Lucy’s lumbar vertebrae were recovered, and they show features consistent with lumbar lordosis—the inward curvature of the lower back that is a hallmark of bipedal posture. This curvature positions the upper body’s center of mass directly above the hips, an essential adaptation for efficient bipedal balance. Chimpanzees lack this curvature; their spines form a single C-shaped arc. Lucy’s vertebrae suggest her lower back was at least partially lordotic—not to the degree seen in modern humans, but clearly differentiated from the ape condition.
The foot, though poorly preserved in Lucy herself, has been illuminated by other A. afarensis specimens. A complete fourth metatarsal recovered from Hadar in 2011 (specimen AL 333-160) shows evidence of both transverse and longitudinal arches—the spring-loaded architecture that stores and releases elastic energy during bipedal walking and running. The foot of A. afarensis was not a flat, grasping appendage like a chimpanzee’s. It was an arched, stiffened lever built for propulsive push-off during the stance phase of bipedal gait.
Anatomical Comparison
D1: Anatomical Comparison Across Species
Key morphological traits compared across Chimpanzee, A. afarensis (Lucy), and Modern Human. Toggle species columns on or off.
| Trait | Unit | Chimpanzee | A. afarensis | Modern Human |
|---|---|---|---|---|
| Cranial capacity | cc | ~393 | ~438 | ~1350 |
| Femoral bicondylar angle | ° | ~0 | ~9 | 9–11 |
| Intermembral index | ~108 | ~88 | ~72 | |
| Pelvic inlet (AP/transverse) | ~1.0 | ~0.74 | ~0.80 | |
| Lumbar lordosis | Absent | Partial | Present | |
| Foot longitudinal arch | Absent | Present | Present | |
| Foramen magnum position | Posterior | Anterior | Anterior | |
| Estimated stature | cm | 100–130 | ~107 | 162–176 |
| Estimated body mass | kg | 32–42 | ~29 | 62–78 |
But the most vivid evidence for afarensis bipedalism comes not from bones at all. It comes from footprints. In 1978, Mary Leakey’s team at Laetoli in Tanzania uncovered a trail of hominin footprints preserved in a layer of volcanic ash dated to approximately 3.66 million years ago. Two individuals—possibly three—had walked across a fresh ashfall from the Sadiman volcano, and their footprints had been preserved when the ash hardened. The prints show a clear heel strike, a longitudinal arch, a medial weight transfer, and a strong big-toe push-off—the signature of human-like bipedal gait. There is no divergent hallux, no evidence of grasping. These are the footprints of a creature that walked the way you walk.
The Laetoli prints are older than Lucy by roughly half a million years, but they are attributed to Australopithecus afarensis on the basis of contemporaneous fossils from the same site. They confirm, from an entirely independent line of evidence, what Lucy’s pelvis and femur had already argued: by at least 3.7 million years ago, hominins were committed bipeds. They walked upright across an open volcanic landscape with a gait that, at the level of footprint morphology, is indistinguishable from our own.

Replica of the Laetoli footprints (3.66 Ma), attributed to Australopithecus afarensis. The prints show a modern-like heel strike, arch, and toe-off pattern—direct evidence of habitual bipedal gait half a million years before Lucy.
Biomechanical Parameters
D10: Bipedalism Biomechanics Comparison
Gait and locomotor parameters across Chimpanzee, A. afarensis, and Modern Human, showing the progressive adaptation to obligate bipedalism.
| Parameter | Chimpanzee | A. afarensis | Modern Human |
|---|---|---|---|
| Femoral bicondylar angle | ~0° | ~9° | 9–11° |
| Pelvic shape | Tall, narrow | Short, broad | Short, broad |
| Knee flexion at midstance | ~40° (bent) | ~10–15° | ~5° (extended) |
| Hip extension range | Limited | Moderate | Full |
| Gluteus medius function | Extensor | Abductor | Abductor |
| Heel strike | Absent | Present | Present |
| Longitudinal foot arch | Absent | Present | Present |
| Stride/leg-length ratio | ~0.7 | ~0.9–1.0 | ~1.2 |
| Relative locomotion cost | ~4× human | ~1.5–2× | 1× (baseline) |
What the biomechanical data makes clear is that Lucy was not an awkward, shuffling biped experimenting with uprightness. Her anatomy represents millions of years of selection for bipedal efficiency. The valgus angle, the abductor-configured pelvis, the anterior foramen magnum, the arched foot, the heel-strike gait pattern preserved at Laetoli—these are not the features of an animal that occasionally stands up. They are the features of an animal that has been walking upright long enough for natural selection to have fine-tuned the entire postcranial skeleton for the task. The remaining questions—and they are significant—concern not whether Lucy walked upright, but how efficiently, how exclusively, and how much time her species still spent in the trees. Those questions would produce some of the most heated debates in paleoanthropology, and they remain unresolved today.
The Locomotion Debate
Or Built to Climb?
The case for Lucy as an upright walker seemed strong — but not everyone was convinced. In 1983, Jack Stern and Randall Susman published a landmark paper that challenged the emerging orthodoxy of obligate bipedalism. Their argument was anatomical and precise: Lucy’s hands and feet told a different story from her pelvis and knees. Her manual phalanges — the finger bones — were curved in a manner typical of primates that spend significant time gripping branches. Her pedal phalanges, the toe bones, showed the same curvature. These were not the straight, flattened digits of an animal that had fully abandoned the trees. They were the fingers and toes of a climber.
The shoulder told a similar story. Lucy’s scapular morphology revealed a cranially oriented glenoid fossa — the socket where the arm bone meets the shoulder blade pointed upward rather than laterally, as in modern humans. This orientation suggests regular overhead arm use, the kind demanded by climbing vertically through a forest canopy. In a creature that only walked on the ground, this feature would be unnecessary, even maladaptive. Stern and Susman argued that it was evidence of a creature that still spent significant time above ground level.
Then there was the question of proportions. Lucy’s intermembral index — the ratio of arm length to leg length — sat at approximately 88. Modern humans, with their elongated legs built for striding, have an index around 72. Chimpanzees, with long arms for swinging and climbing, come in around 108. Lucy fell squarely between them, her limb proportions suggesting a body that was neither fully committed to the ground nor wholly at home in the canopy. She was, Stern and Susman proposed, a facultative biped: an animal that walked upright when traveling on the ground but retained significant arboreal capability.
The debate that followed lasted decades. On one side stood the “terrestrial bipedalists,” led by Owen Lovejoy and Tim White, who argued that the curved phalanges were evolutionary leftovers — retained primitive features that no longer served a functional purpose. On the other side, Stern, Susman, and their allies maintained that natural selection does not preserve energetically expensive anatomy without reason. Curved finger bones require robust flexor muscles. Upward-facing shoulder sockets demand a different configuration of the rotator cuff. These were not vestigial ornaments. They were working adaptations.
The evidence continued to accumulate on both sides. In 2012, David Green and Zeresenay Alemseged brought a new witness into the argument: the Dikika child, known as Selam (DIK-1-1). This remarkably complete juvenile skeleton, dated to approximately 3.3 million years ago, belonged to a young A. afarensis — a child of roughly three years old. Green and Alemseged studied the shoulder blade in detail and found that it resembled a young gorilla’s far more closely than a young human’s. In modern human children, the scapula remodels early in development as the infant begins to use its arms for reaching and manipulation rather than climbing. The Dikika child’s scapula had not made that transition. It was built for the canopy.
Then, in 2016, John Kappelman and his colleagues at the University of Texas performed high-resolution CT scans of Lucy’s original fossils — the first time the bones had been subjected to this level of scrutiny. What they found was startling. Multiple bones showed fracture patterns consistent with a high-energy vertical deceleration event — in plain language, a fall from a considerable height. The proximal humerus was shattered in a pattern that orthopedic surgeons recognized immediately: it was the kind of break that occurs when a person extends their arms to brace against the ground during a fall. The pattern was bilateral, affecting both arms, suggesting a simultaneous impact. Kappelman estimated that Lucy had fallen from a tree at least twelve meters tall.
The finding was controversial — some researchers argued the fractures were post-mortem, caused by millions of years of sediment compression rather than a living fall. But the implication was profound. If Lucy died falling from a tree, she was in a tree. And if she was in a tree, then the facultative biped model was not merely a theoretical possibility but a lived reality. The truth, as it so often does in paleoanthropology, may lie in a synthesis: Lucy was a capable, habitual biped who also slept in trees, foraged in the canopy, and fled predators by climbing. She did not choose between ground and sky. She inhabited both.
The Environment
The Pliocene World
To understand Lucy, you must understand her world — and her world was not ours. At 3.2 million years ago, the Earth was in the midst of the mid-Pliocene warm period, a climatic phase when global temperatures ran roughly 2.5 to 3 degrees Celsius warmer than the present day. Atmospheric carbon dioxide hovered between 380 and 420 parts per million — a range that modern civilization has only recently re-entered after millions of years. The polar ice caps were smaller. Sea levels stood perhaps 15 to 25 meters higher than today. The planet, in its broad strokes, was a warmer and wetter place.
But the broad strokes are deceptive. East Africa in the Pliocene was undergoing a transformation that would ultimately reshape the trajectory of human evolution. The East African Rift System — a tectonic fracture running thousands of kilometers from the Afar Triangle in the north to Mozambique in the south — was actively tearing the continent apart. As the rift widened and its margins rose into highlands, they created a rain shadow that fundamentally altered the climate of the eastern lowlands. Moisture-laden air from the Congo Basin, which had once swept freely across the continent, was now intercepted by rising escarpments. The forests on the eastern side began to thin, fragment, and retreat.
What replaced them was not the open savanna of popular imagination. The Hadar region where Lucy lived was a complex environmental mosaic — a patchwork of habitats that shifted across relatively short distances. Along the river courses, gallery forests persisted, their canopies providing shade, fruit, and refuge from predators. Beyond the riparian corridors, the land opened into grassland floodplains dotted with seasonal wetlands that expanded during the rains and contracted to mudflats during the dry months. Between forest and grassland lay transitional zones of bushland and open woodland, where scattered trees provided both food and emergency escape routes.

The fauna of Lucy’s world reflected this ecological complexity. Ancestral elephants — proboscideans of the genus Deinotherium, with their downward-curving tusks — browsed in the woodland margins. Three-toed horses of the genus Hipparion, smaller and more gracile than their modern relatives, grazed the open plains alongside herds of antelope. Giant pigs, some exceeding the size of modern hippos, rooted through the undergrowth. In the rivers and wetlands, crocodiles waited, some reaching lengths that dwarfed any living species. And in the shadows of the gallery forests, saber-toothed cats of the genus Dinofelis hunted — ambush predators whose elongated canines were designed for puncturing the hides of slow-moving prey.
This was not a landscape that rewarded specialists. It demanded versatility — the ability to find food in multiple habitats, to read shifting seasonal cues, to flee into trees when a predator appeared on the open plain and to cross open ground when the forest offered nothing. It was a world that favored exactly the kind of creature Lucy appears to have been: a generalist, comfortable in the margins between worlds, able to exploit the full breadth of a mosaic environment that was itself in constant flux.

The cooling trend visible in the final stretch of this timeline would accelerate dramatically. By 2.8 million years ago, global temperatures had dropped substantially, carbon dioxide levels were falling toward 280 ppm, and the grasslands of East Africa were expanding at the expense of forest. This cooling would trigger the onset of Northern Hemisphere glaciation and mark the beginning of the Pleistocene ice ages. Lucy’s species, Australopithecus afarensis, would disappear from the fossil record around 2.9 million years ago — right as the world began its long descent into cold.
Diet and Ecology
What She Ate
We cannot watch Lucy eat. We cannot follow her through the gallery forests or observe her scanning the floodplain for edible sedges. But locked within her bones and teeth is a chemical archive that speaks with remarkable precision about what she consumed during her lifetime. The method is called stable carbon isotope analysis, and it exploits a fundamental difference in how plants capture carbon from the atmosphere.
Plants that use the C3 photosynthetic pathway — which includes most trees, shrubs, and forest-floor herbs — preferentially incorporate the lighter carbon-12 isotope, producing tissues with distinctly negative δ¹³C values. Plants that use the C4 pathway — primarily tropical grasses and sedges — discriminate less against the heavier carbon-13, yielding values closer to zero. When an animal eats these plants, the isotopic signature passes into its bones and tooth enamel, preserved for millions of years in the mineral lattice of bioapatite. By measuring the δ¹³C values in fossil teeth, researchers can reconstruct the broad dietary categories of animals that have been dead for geological ages.
The results for Australopithecus afarensis are telling. With a mean δ¹³C value of approximately −8.0 per mil, Lucy’s species falls squarely in the middle of the isotopic spectrum. Pure forest dwellers like chimpanzees, which eat almost exclusively C3 foods — fruits, leaves, bark, and pith — show values around −13 per mil. At the other extreme, Paranthropus boisei, the hyper-robust “Nutcracker Man” of the East African Plio-Pleistocene, records values near −0.5 per mil, indicating a diet overwhelmingly dominated by C4 resources — probably papyrus sedges and grasses. Lucy sits between these poles: she ate mostly forest foods but supplemented with meaningful quantities of resources from open, grassy environments.
Her teeth corroborate this picture. The molars of A. afarensis are large relative to body size, with thick enamel — a combination that resists the wear and fracture associated with hard or tough foods. Dental microwear analysis, which examines the microscopic scratches and pits left on tooth surfaces by the last meals consumed before death, reveals a mixed signal: fine parallel scratches consistent with soft fruit consumption, interspersed with deeper pitting that suggests harder fallback foods like nuts, seeds, or tough underground storage organs such as tubers and corms.
The dietary picture that emerges is one of flexible opportunism. Lucy was not a specialist. She did not rely on a single food source or a single habitat. She moved between forest and open ground, between the canopy and the floodplain, taking what each environment offered as the seasons turned. In the wet months, when the gallery forests bore fruit and the understory was rich with young leaves, she may have spent most of her time in the trees. In the dry season, when the forests contracted and the woodland margins offered little, she ventured onto the open floodplain to dig for tubers and harvest the seeds of C4 grasses.
“The isotopic data suggest A. afarensis was already exploiting open-country resources — but had not abandoned the trees.”Cerling et al., 2013
This dietary flexibility was not trivial. It was, in evolutionary terms, a key adaptation — perhaps as important as bipedalism itself. In a landscape where environments shifted across short distances and where seasonal variation was pronounced, the ability to switch food sources was the difference between survival and extinction. The more specialized hominin species that came later — the massive-jawed Paranthropus lineage that committed so heavily to C4 foods — would ultimately go extinct when their preferred habitats vanished. Lucy’s kind, the generalists, the in-between creatures, would give rise to the lineage that survived.
Sexual Dimorphism and Social Structure
Among Her Kind
Lucy was small. At roughly 107 centimeters tall and an estimated 29 kilograms, she would have stood barely to the chest of a modern adult human. Her bones are gracile, her joints modest, her frame — even by the standards of her species — diminutive. For decades, researchers assumed her small size simply represented what A. afarensis looked like. Then the AL 333 site told a different story.
The AL 333 locality, known informally as the “First Family” site, lies in the same Hadar Formation that yielded Lucy, but in slightly different stratigraphic layers. When Donald Johanson and his team excavated it between 1975 and 1977, they recovered the remains of at least thirteen individual A. afarensis — males, females, and juveniles of various ages, all found within a relatively confined area. The preservation was fragmentary but the sample was unprecedented: for the first time, researchers had enough individuals from a single species, found in a single deposit, to begin asking questions about population-level variation.
What they found was striking. The largest specimens — presumed males on the basis of size and robusticity — suggested individuals standing approximately 151 centimeters tall and weighing perhaps 45 kilograms. This gives a sexual dimorphism ratio of roughly 1.55, calculated as male body mass divided by female body mass. To put that number in context: modern humans have a dimorphism ratio of about 1.26. Chimpanzees are similar, around 1.31. But gorillas — whose massive silverback males can weigh more than twice as much as adult females — register at approximately 2.36. Lucy’s species fell between humans and gorillas, substantially more dimorphic than we are, though not as extreme as the great apes.

What does this level of dimorphism imply about social organization? In living primates, the correlation between body size dimorphism and mating system is well documented, though imperfect. Species with high dimorphism tend toward polygynous mating systems — social arrangements in which a single dominant male maintains reproductive access to multiple females, and male-male competition for mates drives selection for larger male body size. Gorillas are the textbook example: a silverback controls a harem, and his massive frame is both weapon and advertisement. Species with low dimorphism, by contrast, tend toward monogamy or multi-male, multi-female groups where male competition is less intense.
If the dimorphism estimates for A. afarensis are correct, they suggest a social structure closer to the gorilla model than to the human one: small groups organized around a dominant male, with intense male-male competition and limited pair-bonding. But this interpretation has not gone unchallenged. In 2003, Philip Reno and colleagues published a statistical reanalysis of the AL 333 sample and argued that previous studies had overestimated the dimorphism by conflating specimens from different time periods and possibly different species. When they restricted their analysis to the AL 333 assemblage alone, they found dimorphism levels closer to those of modern humans — around 1.2 to 1.3. If Reno is right, the social implications change dramatically: less male competition, possibly more cooperative group structures, perhaps even the earliest glimmers of pair-bonding.
The debate remains unresolved, in part because the fossil sample, while unusually large for paleoanthropology, is still small by the standards of population biology. Thirteen individuals is not a census. The methods used to estimate body mass from fragmentary postcranial bones carry wide margins of error. And the assumption that primate dimorphism maps cleanly onto mating systems has itself come under scrutiny, with some researchers arguing that ecological factors — predation pressure, dietary competition, substrate use — can drive dimorphism independently of mating behavior.
What the AL 333 site does tell us unambiguously is that these individuals died together. The taphonomic evidence — the condition of the bones, their spatial distribution, the sedimentary context — is consistent with a single catastrophic event, most likely a flash flood that swept a group of A. afarensis from the riverbank and buried them in the same deposit. This is direct evidence of group living. Whatever the precise social structure, Lucy did not wander the Pliocene landscape alone. She lived among her kind, in groups that included adults of both sexes and juveniles of various ages. When disaster struck, they were together.
The Fall
For decades after her discovery, the question of how Lucy died remained unanswerable. Fossils preserve anatomy, not biography. Bones record the shape of a life but rarely the manner of its ending. Then, in 2016, John Kappelman and his colleagues at the University of Texas published a paper in Nature that changed the conversation entirely. Using high-resolution computed tomography — scanning Lucy's bones at a resolution finer than a grain of sand — they claimed to have found something no one had seen before: the signature of her death, written into the fractures themselves.
The CT scans revealed compressive fractures across multiple skeletal elements. Both humeri were fractured. The right distal radius was broken. The left proximal femur showed compression at the neck. The first thoracic vertebra, the sacrum, several ribs, the left os coxa, and the right scapula — all bore evidence of traumatic damage. Kappelman's team argued that the pattern was not random. It was not the kind of diffuse, directionless breakage that sediment compression produces over three million years. It was systematic, consistent, and anatomically coherent: the fracture signature of a body that had hit the ground from a great height.
The key evidence lay in the right proximal humerus. The fracture was a sharp, clean, four-part compressive break with bone fragments still preserved in their original position — not displaced, not scattered, not abraded by the slow grinding of geological time. In modern clinical orthopedics, this pattern has a specific name and a specific cause. It is what emergency physicians see when a patient arrives after a fall from a roof or a ladder: the proximal humeral fracture produced when a conscious person throws out their arms to break a fall, and the full force of vertical deceleration drives through the outstretched limb. The humerus shatters not at the weakest point, but at the point of greatest compressive load.
The proposed scenario was startlingly specific. Lucy fell from a height of at least twelve meters — roughly the height of a four-story building, or the canopy of an acacia tree. She may have been sleeping in the branches, as chimpanzees do, or foraging for fruit in the upper canopy. The bilateral nature of the humeral fractures suggested that she extended both arms simultaneously, a reflexive response that implies consciousness at the moment of impact. She was not dead when she hit the ground. She was awake, and she tried to save herself.
Kappelman speculated that Lucy may have survived the initial impact for a brief, terrible interval. The fracture pattern in the pelvis and femur suggested a secondary impact — her body crumpling after the arms failed to absorb the full force. The rib fractures and vertebral compression pointed to thoracic collapse. Death would not have been instantaneous but it would not have been long delayed. She would have lain at the base of the tree, broken and unable to move, her small body folding in on itself.
“We can see that she broke her arm by throwing out her hand to break her fall — a Loss of consciousness and death would have followed swiftly.”— John Kappelman, University of Texas at Austin, 2016
The counter-argument was fierce, and it came from the taphonomists — the scientists who study what happens to bones after death, during the long transit from organism to fossil. Their objection was fundamental. Three point two million years is an almost inconceivable span of time. During those millions of years, Lucy's bones were buried under meters of sediment, compressed by the weight of accumulating rock, saturated with groundwater, fractured by mineral crystallization, and warped by tectonic forces. The taphonomists argued that virtually every fossil hominin ever recovered shows fracture patterns indistinguishable from those Kappelman attributed to a fall. Sediment compression routinely produces sharp, clean, compressive breaks. Post-mortem fracturing can be bilateral. Ribs and scapulae, being thin and fragile, break in almost every specimen regardless of cause of death.
The methodological critique cut deeper still. Critics charged that Kappelman's team had engaged in a form of confirmation bias — selecting the fractures that supported the fall hypothesis while downplaying or ignoring the many fractures that did not fit the narrative. In any fossil as old as AL 288-1, the bones are a palimpsest of damage accumulated over geological time. Cherry-picking from that record to construct a coherent death scene is, the critics argued, not science but storytelling — compelling, evocative, but ultimately unfalsifiable. The debate remains unresolved. The fractures are real. Their meaning is not.
Fracture Analysis: Competing Interpretations
Nine documented fracture sites on AL 288-1. Toggle between the fall hypothesis and the taphonomic counter-argument.
| # | Bone | Fracture Type | Fall Interpretation |
|---|---|---|---|
| 1 | Right proximal humerus | Compressive, 4-part | Outstretched arm impact during vertical fall |
| 2 | Left proximal humerus | Similar compressive | Bilateral pattern = simultaneous impact |
| 3 | Right distal radius | Greenstick-type | Wrist impact (Colles’-like fracture) |
| 4 | Left proximal femur | Femoral neck compression | Vertical deceleration impact |
| 5 | First thoracic vertebra | Compression | Axial loading from vertical impact |
| 6 | Left os coxa | Superior pubic ramus | High-energy pelvic impact |
| 7 | Right scapula | Glenoid fracture | Shoulder-first impact |
| 8 | Sacrum | Multiple fractures | Vertical impact loading |
| 9 | Multiple ribs | Fractures | Thoracic compression from fall |
What is not in dispute is the strangeness of finding a story in stone — of reading, in the fracture lines of a three-million-year-old humerus, the last seconds of an individual life. Whether Lucy fell from a tree or whether her bones were broken by the slow, impersonal weight of time, the very fact that we can ask the question speaks to something remarkable about her preservation, and about the lengths to which science will go to recover a single, fleeting moment from the abyss of deep time.
Her Contemporaries
It is tempting to imagine Lucy alone — a solitary figure on an empty landscape, the sole representative of the human lineage in her time. This is the image that popular accounts have reinforced for decades: a single line of ancestors, each giving rise to the next, a chain of improvement leading inevitably to us. But the fossil record tells a radically different story. Lucy was not alone. Between four million and two million years ago, East and South Africa hosted a remarkable, almost bewildering diversity of hominin species — a proliferation of evolutionary experiments in bipedalism, diet, brain size, and ecological strategy that makes the human family tree look less like a tree and more like a bush.
At 3.5 million years ago — just three hundred thousand years before Lucy lived — at least three hominin species walked the landscapes of eastern Africa simultaneously. Australopithecus afarensis occupied the Ethiopian Rift, ranging from Hadar in the north to Laetoli in present-day Tanzania. But she shared the continent with Kenyanthropus platyops, a flat-faced hominin discovered by Meave Leakey and colleagues at Lomekwi on the western shore of Lake Turkana in 2001. And in 2015, Yohannes Haile-Selassie and his team announced the discovery of yet another species, Australopithecus deyiremeda, from the Woranso-Mille area — just fifty kilometers from Hadar, and dating to the same time period as Lucy's species. Three species of bipedal hominins, living in overlapping ranges, at the same moment in geological time.

The Taung Child — type specimen of Australopithecus africanus, discovered by Raymond Dart in 1924. This juvenile skull from South Africa represents one of several hominin species that lived alongside or shortly after A. afarensis.
Further south, in the limestone caves of the Cradle of Humankind near Johannesburg, Australopithecus africanus had established itself by at least 3.3 million years ago. The Taung Child, discovered by Raymond Dart in 1924, was the first australopithecine ever recognized — decades before Lucy would be found. A. africanus had a slightly larger brain than A. afarensis, a more rounded cranium, and smaller front teeth, but it retained the same basic body plan: small, bipedal, with arms still adapted for climbing. Whether it descended from Lucy's species or represented an independent lineage remains debated.
By 2.5 million years ago, the cast of characters had expanded further. Australopithecus garhi, discovered at the Bouri Formation in Ethiopia's Middle Awash, appeared with a surprising combination of features — large teeth set in a relatively gracile skull, and an association with some of the earliest stone tools and cut-marked animal bones. To the north and east, the robust lineage had begun its own experiment: Paranthropus aethiopicus, the earliest of the so-called robust australopithecines, had evolved massive jaws, enormous molar teeth, and a sagittal crest — a ridge of bone along the skull's midline for anchoring powerful chewing muscles. This was a species built to process tough, fibrous plant foods, a dietary specialist in a world of generalists.
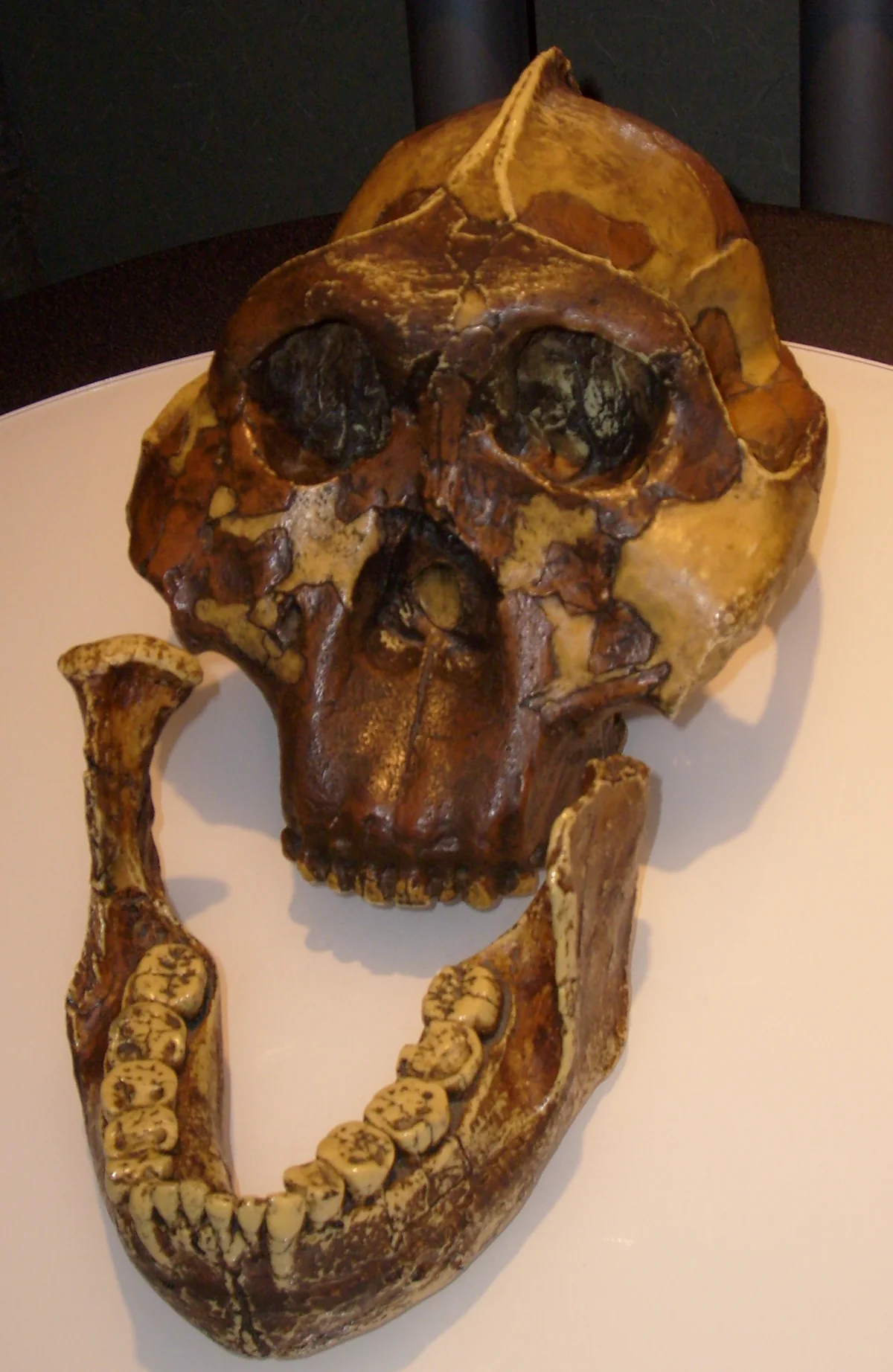
Paranthropus boisei — the hyper-robust “Nutcracker Man.” Its enormous molars and powerful jaw represent one of the most extreme dietary specializations in hominin evolution. It lived alongside early Homo in East Africa between 2.3 and 1.2 million years ago.
And then there was Homo. By 2.8 million years ago, at the site of Ledi-Geraru — less than thirty kilometers from where Lucy was found — a partial mandible designated LD 350-1 records the earliest known member of our own genus. The jaw is slim, the teeth are small, and the chin shows features that foreshadow the direction human evolution would take over the next three million years. The emergence of Homo overlapped with the final populations of A. afarensis. For a brief window, Lucy's descendants — if they were her descendants — walked the same valleys as the first members of the lineage that would eventually produce us.
This is not a ladder. It is not a march of progress. It is a radiation — an adaptive explosion of forms, each exploring a different solution to the challenges of Pliocene Africa. Some invested in massive jaws and specialized diets. Others remained generalists. At least one lineage began to expand its brain and refine its stone tools. Most went extinct. The bush was pruned, again and again, by climate shifts, habitat fragmentation, and competition, until only a single twig remained. We are that twig, and the diversity that surrounded Lucy is a reminder of how contingent our survival was.
Hominin Species Presence, 4.0 – 2.0 Ma
Time bins of 0.25 million years. Filled circles indicate species presence in the fossil record; open circles indicate absence.
| Time Bin (Ma) | A. afarensis | A. africanus | K. platyops | A. deyiremeda | A. garhi | P. aethiopicus | P. boisei | Early Homo |
|---|---|---|---|---|---|---|---|---|
| 4.0–3.75 | ● | ○ | ○ | ○ | ○ | ○ | ○ | ○ |
| 3.75–3.50 | ● | ○ | ● | ● | ○ | ○ | ○ | ○ |
| 3.50–3.25 | ● | ● | ● | ● | ○ | ○ | ○ | ○ |
| 3.25–3.00 | ● | ● | ○ | ○ | ○ | ○ | ○ | ○ |
| 3.00–2.75 | ● | ● | ○ | ○ | ○ | ○ | ○ | ● |
| 2.75–2.50 | ○ | ● | ○ | ○ | ● | ● | ○ | ● |
| 2.50–2.25 | ○ | ● | ○ | ○ | ○ | ● | ● | ● |
| 2.25–2.00 | ○ | ● | ○ | ○ | ○ | ○ | ● | ● |
The species presence table above makes the pattern starkly visible. At 3.5 million years ago, three species coexisted. By 2.5 million years, the number had risen to five or more, spanning from the Ethiopian Rift to the South African caves. The robust australopithecines and early Homo appeared almost simultaneously, emerging from a background of diversity that Lucy's generation helped to seed. The old metaphor of the “missing link” — a single chain with a single gap — is not merely outdated. It is the wrong shape entirely.
Comparative Morphology Radar
Normalized trait profiles (0–100) across six morphological dimensions. Toggle species to compare body plans.
Where She Sits
In 1978, when Donald Johanson and Tim White formally described Australopithecus afarensis, they made a bold claim — one that would define the field for a generation. Lucy's species, they argued, was the last common ancestor of all later hominins. She sat at the trunk of the human family tree, the single stem from which every subsequent branch diverged: the gracile australopithecines, the robust Paranthropus lineage, and our own genus Homo. In their model, A. afarensis was not merely an early relative. She was the root — the species from which all of us, ultimately, descend.
This “Stem Species” model was elegant, parsimonious, and enormously influential. It rested on two pillars. First, A. afarensis was the oldest well-documented hominin at the time of the proposal, predating all other known australopithecines and early Homo by at least several hundred thousand years. Second, her anatomy appeared to be generalized — lacking the extreme specializations that characterized later lineages. She had neither the massive jaws of Paranthropus nor the enlarged brain of Homo. She seemed, in the language of cladistics, to be plesiomorphic — retaining ancestral features from which both specialized lineages could plausibly have evolved. For Johanson, White, and their colleague William Kimbel, this was the strongest kind of phylogenetic argument: the simplest explanation that fit all the available evidence.
But cladistics is a demanding discipline, and not all of the evidence pointed in the same direction. Through the 1990s and early 2000s, a series of detailed morphological analyses began to identify features in A. afarensis that appeared to be derived — specialized traits shared with later australopithecines but absent in early Homo. The shape of the mandibular ramus, certain features of the dental arcade, aspects of the facial architecture — these suggested that A. afarensis might not be the ancestor of Homo at all, but rather a close relative on a parallel branch. Daniel Strait and Frederick Grine published a landmark cladistic analysis in 2004 that placed A. afarensis on its own branch — a cousin to the lineage leading to humans, not a direct ancestor. In this “Side Branch” model, the true ancestor of Homo was an unknown species, perhaps one that has not yet been discovered.
Then the bush got thicker. The discovery of Kenyanthropus platyops in 2001, followed by Australopithecus deyiremeda in 2015, demonstrated that the middle Pliocene hosted far more hominin diversity than anyone had imagined when Johanson and White first proposed their stem-species hypothesis. If three, four, or even five species coexisted between 3.8 and 3.0 million years ago, then the probability of correctly identifying any single one as the ancestor of all later hominins becomes vanishingly small. Bernard Wood and Yohannes Haile-Selassie have been the most prominent advocates of this third view — the “Bush” or “Diversity” model — which holds that the hominin family tree in the middle Pliocene was a radiation, not a sequence, and that our attempts to draw clean lines of descent through it may reflect our desire for narrative order more than the biological reality.
Phylogenetic Position of A. afarensis
Three competing models for Lucy’s place in the hominin family tree. Select a model to view its proposed topology.
The debate is not merely academic. The question of where A. afarensis sits in our family tree is, at root, a question about the nature of human evolution itself. If the Stem Species model is correct, then evolution was relatively orderly — a single trunk giving rise to branches in a pattern we can, in principle, reconstruct. If the Bush model is correct, then our origins were chaotic, contingent, and far more complex than any simple tree can represent. The truth almost certainly lies somewhere in the deeply uncomfortable middle: we know enough to see the complexity, but not yet enough to resolve it.
What is remarkable is that all three models agree on one point. Whatever her exact phylogenetic position, Australopithecus afarensis occupies a critical zone in hominin evolution — the interval between 4.0 and 3.0 million years ago when the fundamental hominin body plan was established. She walked upright. She had small canines and a parabolic dental arcade. She lived in social groups. She exploited a range of habitats from woodlands to open floodplains. Whether she was our direct ancestor or our evolutionary cousin, she represents the kind of animal from which all later hominins — including us — emerged. Her significance does not depend on a line drawn on a cladogram. It depends on what she was.
Dinkinesh
In Ethiopia, Lucy is not called Lucy. She is Dinkinesh — an Amharic word that means “you are marvelous.” The name was given to her by the Ethiopian members of the expedition team, and it captures something that the catalogue number AL 288-1 cannot: the astonishment of encountering a creature who lived 3.2 million years ago and who, against every statistical probability, left behind enough of herself to be known.
Lucy is the most famous fossil in the world. This is not because she is the oldest hominin — Ardipithecus ramidus, at 4.4 million years, predates her by more than a million years, and Sahelanthropus tchadensis may reach back nearly seven million. It is not because she is the most complete — the Ardipithecus skeleton and the astonishing StW 573 specimen from Sterkfontein both preserve more of the skeleton than Lucy does. She is famous because she arrived at exactly the right moment in the history of paleoanthropology to transform the field, and because she arrived with a story that the public could understand.

A life reconstruction of Australopithecus afarensis, based on the skeletal evidence from AL 288-1 and other Hadar specimens. She stood roughly 107 centimeters tall and weighed about 29 kilograms — small by any human standard, but fully committed to a bipedal life.
Before Lucy, the reigning paradigm in human evolution held that the brain led the way. The so-called “cerebral Rubicon” hypothesis, inherited from the early twentieth century, assumed that what made humans human was intelligence — that our ancestors first developed large brains, and that bipedalism, tool use, and all the other hallmarks of humanity followed as consequences of cognitive superiority. This was a deeply flattering narrative. It placed the mind at the center of human evolution and made everything else secondary.
Lucy destroyed that narrative. Her brain was approximately 438 cubic centimeters — barely larger than a chimpanzee's, firmly within the range of a great ape, and less than a third the size of a modern human brain. Yet her pelvis was unmistakably adapted for bipedal locomotion. Her femur angled inward at the knee. Her foot had a longitudinal arch. She walked upright — not clumsily, not tentatively, but as her primary mode of locomotion — with a brain no larger than a softball. The inversion was total and irreversible. Bipedalism came first. The brain came later — at least two million years later. Our ancestors were upright walkers long before they were thinkers, and Lucy was the proof.
This insight did more than revise a scientific hypothesis. It reshaped how the public understood what it means to be human. Before Lucy, human evolution was an abstraction — a subject for specialists, illustrated by fragments of skull and dense jargon. Lucy gave the story a protagonist. She had a name. She had a height (107 centimeters), a weight (about 29 kilograms), and an estimated age (young adult). She had a sex, a species, and — if Kappelman is right — a death. She was not a diagram or a phylogenetic node. She was an individual, and the public responded to her with an intensity that no fossil had ever provoked.
When Lucy toured American museums in the mid-2000s, hundreds of thousands of people lined up to see her — not a cast, but the actual bones, 3.2 million years old, laid out under glass. Ethiopian officials debated fiercely whether the bones should travel at all, and the controversy itself underscored how deeply Lucy had been woven into Ethiopian national identity. She belongs to Ethiopia. She was found in Ethiopian soil, she is housed in the Ethiopian National Museum in Addis Ababa, and her Amharic name — Dinkinesh — is the one her discoverers heard first.
In 2023, NASA launched a spacecraft to study the Trojan asteroids of Jupiter. They named it Lucy — after the fossil, who was named after the Beatles song, which was playing in camp on the night of her discovery. The mission patch includes a diamond, a nod to the song's title. It is a strange and beautiful chain of references: from a Pliocene hominin in the Afar Triangle, to a pop song recorded in Abbey Road Studios in 1967, to a robotic explorer sailing past the orbit of Mars. The connections are arbitrary and human, which is to say they are exactly the kind of meaning-making that distinguishes us from every other primate — and from the small, long-dead creature who helped us understand why.
“She is 3.2 million years old, and she has changed the way we think about ourselves. She showed us that we walked before we thought — that the human story begins not with the mind, but with the body, moving across a landscape, upright and free.”— Donald Johanson
Dinkinesh. You are marvelous. A small body, a small brain, a set of bones scattered across a hillside in the Afar depression, and a legacy that reaches from the deep Pliocene to the outer solar system. She is a bridge between the alien world of deep time and the intimate reality of a single life lived and lost — three million years of silence, broken by the sound of a knee joint being lifted, for the first time, out of the sediment.
Sources & Further Reading
- 1.Johanson, D.C. & White, T.D. (1979). A systematic assessment of early African hominids. Science, 203(4378), 321–330.
- 2.Johanson, D.C. & Edey, M.A. (1981). Lucy: The Beginnings of Humankind. Simon & Schuster.
- 3.Kimbel, W.H. & Delezene, L.K. (2009). “Lucy” redux: A review of research on Australopithecus afarensis. American Journal of Physical Anthropology, 140(S49), 2–48.
- 4.Kappelman, J. et al. (2016). Perimortem fractures in Lucy suggest mortality from fall out of tall tree. Nature, 537, 503–507.
- 5.Stern, J.T. Jr. & Susman, R.L. (1983). The locomotor anatomy of Australopithecus afarensis. American Journal of Physical Anthropology, 60(3), 279–317.
- 6.Lovejoy, C.O. (1988). Evolution of human walking. Scientific American, 259(5), 118–125.
- 7.Ward, C.V., Kimbel, W.H. & Johanson, D.C. (2011). Complete fourth metatarsal and arches in the foot of Australopithecus afarensis. Science, 331(6018), 750–753.
- 8.Leakey, M.G. et al. (2001). New hominin genus from eastern Africa shows diverse middle Pliocene lineages. Nature, 410(6827), 433–440.
- 9.Haile-Selassie, Y. et al. (2015). New species from Ethiopia further expands Middle Pliocene hominin diversity. Nature, 521(7553), 483–488.
- 10.Wood, B. & Boyle, E.K. (2016). Hominin taxic diversity: Fact or fantasy? American Journal of Physical Anthropology, 159(S61), 37–78.
- 11.Cerling, T.E. et al. (2013). Stable isotope-based diet reconstructions of Turkana Basin hominins. Proceedings of the National Academy of Sciences, 110(26), 10501–10506.
- 12.Reed, K.E. (1997). Early hominid evolution and ecological change through the African Plio-Pleistocene. Journal of Human Evolution, 32(2–3), 289–322.
- 13.Wynn, J.G. et al. (2006). Geological and palaeontological context of a Pliocene juvenile hominin at Dikika, Ethiopia. Nature, 443(7109), 332–336.
- 14.McHenry, H.M. (1992). Body size and proportions in early hominids. American Journal of Physical Anthropology, 87(4), 407–431.
- 15.Reno, P.L. et al. (2003). Sexual dimorphism in Australopithecus afarensis was similar to that of modern humans. Proceedings of the National Academy of Sciences, 100(16), 9404–9409.
- 16.Villmoare, B. et al. (2015). Early Homo at 2.8 Ma from Ledi-Geraru, Afar, Ethiopia. Science, 347(6228), 1352–1355.
- 17.Alemseged, Z. et al. (2006). A juvenile early hominin skeleton from Dikika, Ethiopia. Nature, 443(7109), 296–301.
- 18.Holloway, R.L. et al. (2004). The Human Fossil Record, Volume 3: Brain Endocasts. Wiley-Liss.
- 19.Dart, R.A. (1925). Australopithecus africanus: The man-ape of South Africa. Nature, 115(2884), 195–199.
- 20.White, T.D. et al. (2009). Ardipithecus ramidus and the paleobiology of early hominids. Science, 326(5949), 64–86.
- 21.Leakey, L.S.B. (1959). A new fossil skull from Olduvai. Nature, 184(4685), 491–493.
- 22.Green, D.J. & Alemseged, Z. (2012). Australopithecus afarensis scapular ontogeny, function, and the role of climbing in human evolution. Science, 338(6106), 514–517.
- 23.Haile-Selassie, Y. et al. (2010). An early Australopithecus afarensis postcranium from Woranso-Mille, Ethiopia. Proceedings of the National Academy of Sciences, 107(27), 12121–12126.
- 24.Latimer, B. & Lovejoy, C.O. (1989). The calcaneus of Australopithecus afarensis and its implications for the evolution of bipedality. American Journal of Physical Anthropology, 78(3), 369–386.
Image Credits
- Lucy skeleton (National Museum, Mexico City) — Museo Nacional de Antropología, CC BY-SA 2.0
- Lucy skeleton (articulated) — 120, CC BY-SA 3.0
- Lucy skeleton (black background) — John Gurche / Cleveland Museum of Natural History, CC BY-SA 3.0
- Lucy reconstruction — James St. John, CC BY 2.0
- A. afarensis skull (Senckenberg Museum) — Daderot, CC0 1.0
- A. afarensis reconstruction — Cicero Moraes, CC BY-SA 3.0
- Laetoli footprints replica — Tim Evanson, CC BY-SA 2.0
- Taung Child skull — Didier Descouens, CC BY-SA 4.0
- Paranthropus boisei skull — Nrkpan, CC BY-SA 3.0
- Hadar, Ethiopia — Wikimedia Commons, Public Domain
- Afar Triangle political map — Sémhur, CC BY-SA 3.0
- Afar Depression (ASTER satellite) — NASA/GSFC/METI/ERSDAC/JAROS, Public Domain
- Dallol landscape, Afar — A.Savin, CC BY-SA 3.0
- Lake Karum area, Afar — A.Savin, CC BY-SA 3.0
All images sourced from Wikimedia Commons under Creative Commons or Public Domain licenses. Anatomical and isotopic data drawn from cited primary literature.
Further Reading
- Turkana Boy: The Skeleton That Redefined Human Evolution — KNM-WT 15000, the most complete early human skeleton ever found. An interactive visual essay on the 1.5-million-year-old Homo erectus youth with 3D fossil specimens from the Turkana Basin.
- Homo naledi: The Small-Brained Species That Buried Its Dead — 1,500 bones, 560 cubic centimetres, and the question of what makes us human. The Rising Star Cave discovery that challenges the link between brain size and behavioural complexity.
- Seven Million Years: The Complete Visual History of Our Kind — From Sahelanthropus to Homo sapiens — the complete arc of hominin evolution in one visual essay.
- Infographic: How Our Brains Grew Over 7 Million Years — Cranial capacity comparison across 8 hominid species, from Sahelanthropus (350 cc) to Homo sapiens (1,350 cc).